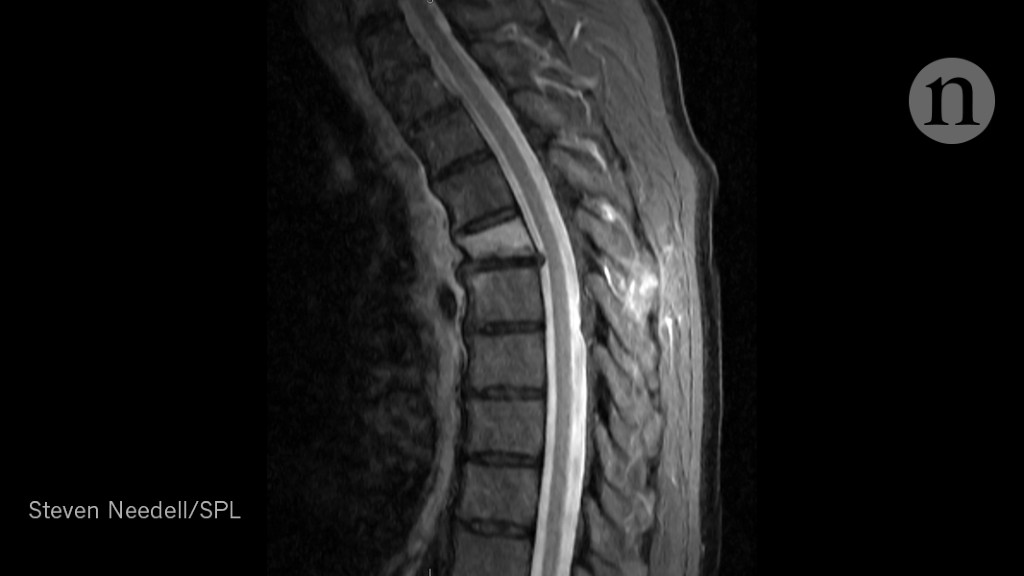
Randomized controlled multicenter trials are warranted. Therefore, when combined with physical therapy, autologous adherent bone marrow cell therapy appears to be a safe and promising therapy for patients with chronic spinal cord injuries. Compared to those patients with cervical injuries, a higher rate of functional improvement was achieved in thoracic SCI patients with shorter durations of injury and smaller cord lesions. At 18 months posttreatment, 23 of the 50 cell therapy-treated cases (46 percent) showed sustained improvement using the American Spinal Injury Association (ASIA) Impairment Scale (AIS). Chronic cervical and thoracic SCI patients treated with autologous adherent bone marrow cells combined with physical therapy showed functional improvements over patients in the control group treated with physical therapy alone, and there were no cell therapy-related side effects. The potential mechanisms of stem cell therapy for SCI include tissue repair and replacement, neurotrophy, regeneration, promotion of angiogenesis, antiapoptosis, and anti-inflammatory. The mechanisms of stem cell (SC) therapy for spinal cord injury (SCI). Patients were evaluated with clinical examinations, electrophysiological somatosensory evoked potential, MRI imaging, and functional independence measurements. The mechanisms of SC treatment of SCI are shown in Figure 2.
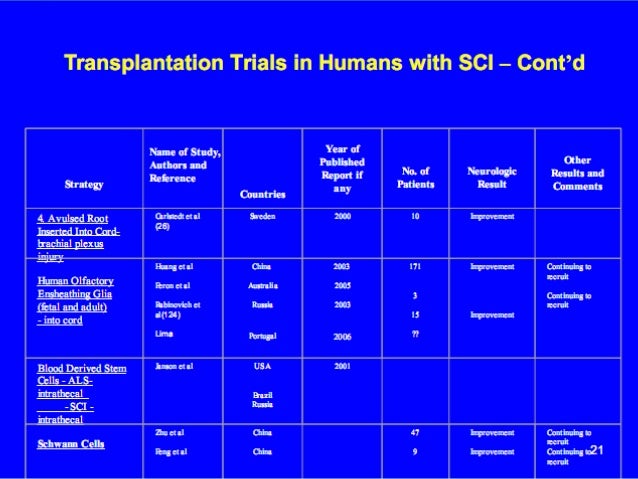
#Stem cell treatment for spinal cord injury trial#
In a phase I/II controlled single-blind clinical trial ( identifier: NCT00816803), 70 chronic cervical and thoracic SCI patients with injury durations of at least 6 months were treated with either intrathecal injection(s) of autologous adherent bone marrow cells combined with physical therapy, or with physical therapy alone. Here we describe a strategy in which treatment with autologous adherent bone marrow cells is combined with physical therapy to improve motor and sensory functions in early-stage chronic SCI patients. Copyright © 2009 John Wiley & Sons, Ltd.Spinal cord injuries (SCI) cause sensory loss and motor paralysis and are treated with physical therapy, but most patients fail to recover due to limited neural regeneration. This study reports the first successful differentiation of MNs from human spinal cord stem cells in vitro, providing a novel approach for obtaining functional MNs when designing the therapeutic strategy for spinal cord diseases or injuries. The recordings of spontaneous excitatory postsynaptic currents (EPSCs) confirmed the formation of synapses onto these MNs. The majority of these MNs fired repetitive action potentials (APs), which is an indicator of functional maturation. Finally, the electrophysiological properties of these differentiated MNs were characterized to confirm their functional integrity. The presence of other cell types in the differentiated culture was also analysed. (2013) Role of Autologous Bone Marrow Mononuclear Cells in Chronic Cervical Spinal Cord Injury-A. The combination of HB9 and ChAT immunostainings indicated that ∼20% of the cells were MNs after this induction protocol. Sharma A, Sane H, Gokulchandran N, Kulkarni P, Thomas N, et al.
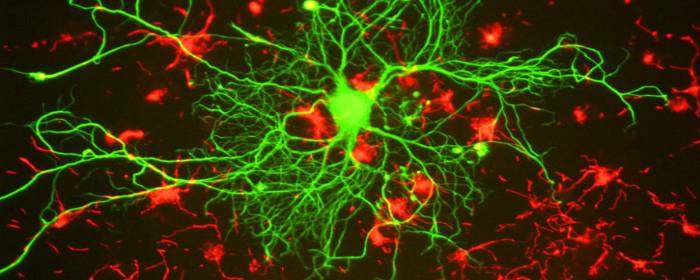
Then a paradigm was introduced to successfully induce MNs from this cell line, which was demonstrated by immunostaining using the MN markers HB9, Islet1 and choline acetyl transferase (ChAT). This study first characterized a polyclonal spinal cord stem cell line isolated from an 8 week-old fetus. Although neural progenitors from spinal cord would be a likely source for generating MNs, there has been no study reporting successful in vitro differentiation of MNs from spinal cord progenitors. So far, in vitro, MNs have only been successfully induced from embryonic stem cells (hESC) and human fetal cortical progenitors.
Specific neuronal subtypes, especially motoneurons (MNs), derived from human stem cells provide a significant therapeutic potential for spinal cord diseases, such as amyotrophic lateral sclerosis (ALS) and spinal cord injury.
